Cytokinetics Presents New Data Related to Aficamten at the European Society of Cardiology Congress 2025
Cytokinetics (NASDAQ:CYTK) presented new data for aficamten at ESC Congress 2025, strengthening its position as a potential treatment for obstructive hypertrophic cardiomyopathy (oHCM). Key findings from the MAPLE-HCM study showed aficamten's superiority over metoprolol in improving cardiac structure and function.
The data revealed that aficamten demonstrated a low annual incidence rate of atrial fibrillation at 1.5%, consistent with expected rates in HCM patients. Long-term safety data from FOREST-HCM showed sustained benefits across 352 patient-years of exposure, with significant improvements in cardiac function and patient symptoms. Notably, 93% of patients reported improvement in NYHA Functional Class at Week 96.
Aficamten is currently under FDA review with a PDUFA date of December 26, 2025.
Cytokinetics (NASDAQ:CYTK) ha presentato nuovi dati su aficamten al Congresso ESC 2025, rafforzando la sua posizione come possibile trattamento per la cardiomiopatia ipertrofica ostruttiva (oHCM). I risultati principali dello studio MAPLE-HCM hanno mostrato la superiorità di aficamten rispetto a metoprololo nel migliorare la struttura e la funzione cardiaca.
I dati hanno rivelato che aficamten ha mostrato un basso tasso di incidenza annua di fibrillazione atriale, pari all'1,5%, coerente con i tassi attesi nei pazienti con HCM. I dati di sicurezza a lungo termine provenienti da FOREST-HCM hanno evidenziato benefici sostenuti su 352 paziente-anni di esposizione, con miglioramenti significativi nella funzione cardiaca e nei sintomi dei pazienti. In particolare, il 93% dei pazienti ha riportato un miglioramento della classe funzionale NYHA alla settimana 96.
Aficamten è attualmente in valutazione presso la FDA con una data PDUFA del 26 dicembre 2025.
Cytokinetics (NASDAQ:CYTK) presentó nuevos datos sobre aficamten en el Congreso ESC 2025, reforzando su posición como posible tratamiento para la miocardiopatía hipertrófica obstructiva (oHCM). Los resultados clave del estudio MAPLE-HCM mostraron la superioridad de aficamten frente a metoprolol en la mejora de la estructura y función cardíaca.
Los datos revelaron que aficamten mostró una baja incidencia anual de fibrilación auricular del 1,5%, coherente con las tasas esperadas en pacientes con HCM. Los datos de seguridad a largo plazo de FOREST-HCM mostraron beneficios sostenidos a lo largo de 352 años-paciente de exposición, con mejoras significativas en la función cardíaca y los síntomas de los pacientes. Cabe destacar que el 93% de los pacientes informó mejoría en la clase funcional NYHA en la semana 96.
Aficamten está actualmente bajo revisión de la FDA con una fecha PDUFA del 26 de diciembre de 2025.
Cytokinetics (NASDAQ:CYTK)�� ESC Congress 2025에서 aficamten�� 대�� 새로�� 데이터를 발표하며 폐쇄�� 비후�� 심근병증(oHCM) 치료 후보로서�� 입지�� 강화했습니다. MAPLE-HCM 연구�� 주요 결과�� aficamten�� 심장 구조와 기능 개선에서 메토프로롤보�� 우수함을 보여주었습니��.
데이터에 따르�� aficamten은 연간 심방세동 발생률이 1.5%�� 낮게 나타��으며, 이는 HCM 환자에서 예상되는 비율�� 일치합니��. FOREST-HCM�� 장기 안전�� 데이터는 352 환자-�� 노출 동안 지속적�� 이점�� 보였��, 심장 기능�� 환자 증상에서 유의�� 개선�� 확인했습니다. 특히 96주차�� 93%�� 환자가 NYHA 기능 분류 개선�� 보고했습니다.
Aficamten은 현재 FDA 심사 중이�� PDUFA 기한은 2025�� 12�� 26��입니��.
Cytokinetics (NASDAQ:CYTK) a présenté de nouvelles données sur l'aficamten au Congrès ESC 2025, renforçant sa position en tant que traitement potentiel de la cardiomyopathie hypertrophique obstructive (oHCM). Les résultats clés de l'étude MAPLE-HCM ont montré la supériorité de l'aficamten par rapport au métoprolol pour l'amélioration de la structure et de la fonction cardiaques.
Les données ont révélé que l'aficamten présentait un faible taux d'incidence annuel de fibrillation atriale de 1,5%, conforme aux taux attendus chez les patients HCM. Les données de sécurité à long terme de FOREST-HCM ont montré des bénéfices durables sur 352 années-patients d'exposition, avec des améliorations significatives de la fonction cardiaque et des symptômes des patients. Notamment, 93% des patients ont rapporté une amélioration de la classe fonctionnelle NYHA à la semaine 96.
L'aficamten est actuellement en cours d'examen par la FDA avec une date PDUFA fixée au 26 décembre 2025.
Cytokinetics (NASDAQ:CYTK) stellte auf dem ESC-Kongress 2025 neue Daten zu Aficamten vor und stärkte damit seine Position als potenzielle Behandlung der obstruktiven hypertrophen Kardiomyopathie (oHCM). Wichtige Ergebnisse der MAPLE-HCM-Studie zeigten die Überlegenheit von Aficamten gegenüber Metoprolol hinsichtlich Verbesserung von Herzstruktur und -funktion.
Die Daten zeigten, dass Aficamten eine
Aficamten befindet sich derzeit in der FDA-Prüfung mit einem PDUFA-Datum am 26. Dezember 2025.
- Aficamten demonstrated superiority over standard-of-care metoprolol in cardiac structure and function improvements
- 93% of patients showed NYHA Functional Class improvement at Week 96
- Low 1.5% annual incidence rate of atrial fibrillation, consistent with expected rates
- Significant improvements in NT-proBNP and cardiac troponin I biomarkers (p<0.0001)
- Strong safety profile with 352 patient-years of exposure and minimal LVEF reduction
- 1.1% of patients experienced transient left ventricular ejection fraction <50%
- 10 patients (3.4%) required dose down-titration due to LVEF <50%
- One patient (0.3%) terminated treatment due to ischemic colitis
Insights
Aficamten shows superior efficacy over standard therapy with favorable safety profile, strengthening its regulatory approval prospects for December 2025.
The new data for Cytokinetics' aficamten provides compelling evidence of superiority over metoprolol, the current standard of care for obstructive hypertrophic cardiomyopathy (oHCM). The MAPLE-HCM trial demonstrates aficamten significantly improves cardiac structure and function with superior diastolic function and reduced mitral valve obstruction - the primary mechanism behind left ventricular outflow tract obstruction in HCM patients.
Particularly noteworthy is the safety profile data addressing two critical concerns: atrial fibrillation risk and ejection fraction reduction. The annual incidence rate of new-onset atrial fibrillation with aficamten was only
The longer-term FOREST-HCM data reinforces aficamten's durability of effect, with sustained hemodynamic improvements through treatment up to three years. The mean reduction in Valsalva LVOT gradient of
With an integrated safety analysis showing adverse event profiles similar to placebo across nearly
Additional Data from MAPLE-HCM Shows Aficamten Improves Cardiac Structure and Function Compared to Metoprolol; Simultaneous Publication in the Journal of the American College of Cardiology
New Analysis Shows Annual Incidence Rate of Atrial Fibrillation with Aficamten is
Longer-Term Data Presented Consistent with Previously Reported Safety Profile of Aficamten
Company to Host Investor Event and Webcast Tuesday September 2, 2025, at 8:30 AM Eastern Time
SOUTH SAN FRANCISCO, Calif., Aug. 31, 2025 (GLOBE NEWSWIRE) -- Cytokinetics, Incorporated (Nasdaq: CYTK) today announced that additional data related to ���ھ��������ٱ����were presented at the European Society of Cardiology Congress 2025 in Madrid, Spain, including a pre-specified analysis of the effect of aficamten on cardiac structure and function from MAPLE-HCM (Metoprolol vs Aficamten in Patients with LVOT Obstruction on Exercise Capacity in HCM), which was simultaneously published in the Journal of the American College of Cardiology1, as well as a Late Breaking Clinical Science presentation related to the incidence and impact of atrial fibrillation (AF) associated with aficamten, which was simultaneously published in Heart Rhythm.2
“The echocardiographic results from MAPLE-HCM elaborate on the superiority of aficamten compared to the current standard-of-care metoprolol as was previously reported in the primary efficacy analyses,�� said Fady I. Malik, M.D., Ph.D., Cytokinetics�� Executive Vice President of Research & Development. “The additional data presented at the ESC Congress 2025 continue to strengthen the evidence base supporting the overall safety profile of aficamten, including as has been observed with a real-world dosing strategy.��
Aficamten is an investigational drug candidate currently under regulatory review in the U.S.; the Food and Drug Administration (FDA) is reviewing a New Drug Application (NDA) for aficamten with a Prescription Drug User Fee Act (PDUFA) target action date of December 26, 2025.
Additional Analysis from MAPLE-HCM Assesses Effect of Aficamten Compared to Metoprolol on Cardiac Structure and Function
Dr. Sheila Hegde, M.D., M.P.H., Assistant Professor, UT Southwestern Medical Center, Dallas, TX, Affiliate Faculty, Brigham and Women's Hospital, Boston, MA, presented a pre-specified echocardiographic analysis assessing the effect of treatment with aficamten relative to metoprolol on cardiac structure and function from MAPLE-HCM.
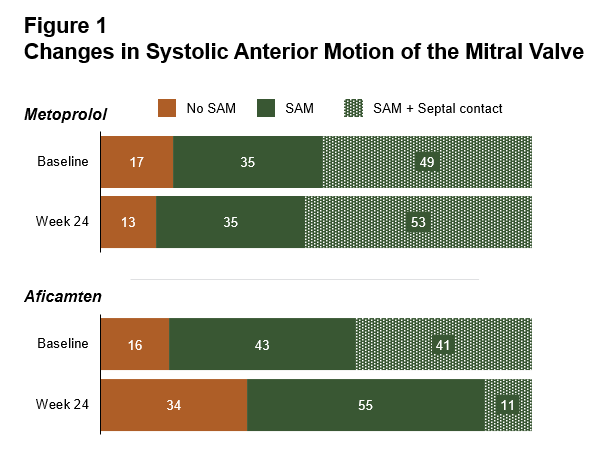
In addition to primary results of MAPLE-HCM presented by Dr. Garcia Pavia at the European Society of Cardiology Congress 2025, aficamten was also shown to be superior to metoprolol at improving measures of diastolic function and reducing the likelihood of mitral valve systolic anterior motion (SAM) and mitral valve leaflet-septal contact, the primary contributor to left ventricular outflow tract (LVOT) obstruction (Figure 1) (all p<0.001). There was a significant decrease in maximal wall thickness in patients treated with aficamten compared to those treated with metoprolol (treatment corrected difference = -1.01 mm [
Long-Term Treatment with Aficamten Does Not Appear to Increase Risk of Atrial Fibrillation in Patients with Obstructive Hypertrophic Cardiomyopathy
Ahmad Masri, M.D., MS, Director of the Hypertrophic Cardiomyopathy Center at Oregon Health & Science University, presented a new analysis of the incidence and impact of AF in patients with obstructive hypertrophic cardiomyopathy (oHCM) in a Late Breaking Clinical Science Session outlining the experience of patients treated with aficamten in the open-label study, FOREST-HCM (Follow-up, Open-Label, Research Evaluation of Sustained Treatment with Aficamten in HCM). These results were also simultaneously published in Heart Rhythm.2
Among 173 patients followed for ��48 weeks, 136 had no history of AF at baseline. Four (
New Data from FOREST-HCM on the Longer-Term Safety and Efficacy of Treatment with Aficamten Consistent with Previous Reports
Sara Saberi, M.D., M.S., Associate Professor of Internal Medicine, University of Michigan Health Frankel Cardiovascular Center, presented new data related to the safety and long-term use of aficamten in FOREST-HCM.
FOREST-HCM, the open-label extension clinical study, enrolled 296 patients with oHCM from May 28, 2021, through the data cutoff of August 31, 2024. The total cumulative exposure to aficamten was 352 patient-years over a mean follow-up of 62 weeks and maximum follow-up of 170 weeks. Consistent with previously presented data from FOREST-HCM, this updated data set demonstrates that long-term treatment with aficamten is associated with early and sustained hemodynamic and clinical benefits, along with a low incidence of new-onset AF and LVEF <
Treatment with aficamten resulted in a significant reduction from baseline in site-read Valsalva LVOT-G by Week 12 (resting LVOT-G = -40 mmHg [-42, -39] and Valsalva LVOT-G = -56 mmHg [
Long-term treatment with aficamten was also associated with reduced symptom burden. At Week 12 (n= 283) and Week 96 (n=44),
One patient (
Updated Integrated Safety Analysis, Including MAPLE-HCM, Provides Additional Evidence on the Safety and Tolerability of Aficamten in oHCM
A new integrated safety analysis from three clinical trials of aficamten presented by Dr. Masri provided additional safety and tolerability evidence. The analysis included data from the Phase 2 clinical trial, REDWOOD-HCM, two Phase 3 clinical trials, SEQUOIA-HCM and MAPLE-HCM, and the open-label extension clinical study, FOREST-HCM.
In this integrated safety analysis of 463 participants with oHCM and almost 700 patient-years of exposure, aficamten was shown to be well-tolerated and had an adverse event profile similar to placebo. There remains a low incidence of LVEF <
Investor Webcast Information
Cytokinetics will host an investor webcast on September 2, 2025, at 8:30 AM Eastern Time to discuss the primary results from MAPLE-HCM and other data presented at the European Society of Cardiology Congress 2025. Interested parties can register online at . The live webcast will be available on the Investors & Media section of the Cytokinetics website at . A replay of the webcast will be archived on the Cytokinetics website for six months.
About Aficamten
Aficamten is an investigational selective, small molecule cardiac myosin inhibitor discovered following an extensive chemical optimization program that was conducted with careful attention to therapeutic index and pharmacokinetic properties.3 Aficamten was designed to reduce the number of active actin-myosin cross bridges during each cardiac cycle and consequently suppress the myocardial hypercontractility that is associated with HCM. In preclinical models, aficamten reduced myocardial contractility by binding directly to cardiac myosin at a distinct and selective allosteric binding site, thereby preventing myosin from entering a force producing state.
The development program for aficamten is assessing its potential as a treatment that improves exercise capacity as measured by peak oxygen uptake (pVO2) and relieves symptoms in patients with HCM. Aficamten was evaluated in SEQUOIA-HCM, a positive pivotal Phase 3 clinical trial in patients symptomatic obstructive hypertrophic cardiomyopathy (HCM). Aficamten received Breakthrough Therapy Designation for the treatment of symptomatic HCM from the U.S. Food & Drug Administration (FDA) and for the treatment of symptomatic obstructive HCM from the National Medical Products Administration (NMPA) in China.
Aficamten is also currently being evaluated in ACACIA-HCM, a Phase 3 clinical trial of aficamten in patients with non-obstructive HCM; CEDAR-HCM, a clinical trial of aficamten in a pediatric population with oHCM; and FOREST-HCM, an open-label extension clinical study of aficamten in patients with HCM.
This communication contains a summary of new data related to the clinical development of aficamten presented at the European Society of Cardiology 2025 Congress. Aficamten is an investigational drug and is not approved by any regulatory agency. Its safety and efficacy have not been established. Aficamten is currently under regulatory review in the U.S., where the FDA is reviewing a New Drug Application (NDA) for aficamten with a Prescription Drug User Fee Act (PDUFA) target action date of December 26, 2025. Additionally, the European Medicines Agency (EMA) is reviewing a Marketing Authorization Application (MAA) for aficamten, and The Center for Drug Evaluation (CDE) of the China National Medical Products Administration (NMPA) is reviewing an NDA for aficamten with Priority Review.
About Hypertrophic Cardiomyopathy
Hypertrophic cardiomyopathy (HCM) is a disease in which the heart muscle (myocardium) becomes abnormally thick (hypertrophied). The thickening of cardiac muscle leads to the inside of the left ventricle becoming smaller and stiffer, and thus the ventricle becomes less able to relax and fill with blood. This ultimately limits the heart’s pumping function, resulting in reduced exercise capacity and symptoms including chest pain, dizziness, shortness of breath, or fainting during physical activity. HCM is the most common monogenic inherited cardiovascular disorder, with approximately 280,000 patients diagnosed, however, there are an estimated 400,000-800,000 additional patients who remain undiagnosed in the U.S.4,5,6 Two-thirds of patients with HCM have obstructive HCM (oHCM), where the thickening of the cardiac muscle leads to left ventricular outflow tract (LVOT) obstruction, while one-third have non-obstructive HCM (nHCM), where blood flow isn’t impacted, but the heart muscle is still thickened. People with HCM are at high risk of also developing cardiovascular complications including atrial fibrillation, stroke and mitral valve disease.7 People with HCM are at risk for potentially fatal ventricular arrhythmias and it is one of the leading causes of sudden cardiac death in younger people or athletes.8 A subset of patients with HCM are at high risk of progressive disease leading to dilated cardiomyopathy and heart failure necessitating cardiac transplantation.
About Cytokinetics
Cytokinetics is a specialty cardiovascular biopharmaceutical company, building on its over 25 years of pioneering scientific innovations in muscle biology, and advancing a pipeline of potential new medicines for patients suffering from diseases of cardiac muscle dysfunction. Cytokinetics is readying for potential regulatory approvals and commercialization of aficamten,��a cardiac myosin inhibitor, following positive results from SEQUOIA-HCM, the pivotal Phase 3 clinical trial in patients with obstructive hypertrophic cardiomyopathy (HCM). Aficamten is also being evaluated in additional clinical trials enrolling patients with obstructive and non-obstructive HCM. In addition,��Cytokinetics is developing omecamtiv mecarbil, a cardiac myosin activator, in patients with heart failure with severely reduced ejection fraction (HFrEF), ulacamten, a cardiac myosin inhibitor with a mechanism of action distinct from aficamten,��for the potential treatment of heart failure with preserved ejection fraction (HFpEF) and CK-089, a fast skeletal muscle troponin activator with potential therapeutic application to a specific type of muscular dystrophy and other conditions of impaired skeletal muscle function.
For additional information about Cytokinetics, visit and follow us on , , and .
Forward-Looking Statements
This press release contains forward-looking statements for purposes of the Private Securities Litigation Reform Act of 1995 (the "Act"). Cytokinetics disclaims any intent or obligation to update these forward-looking statements and claims the protection of the Act's Safe Harbor for forward-looking statements. Examples of such statements include, but are not limited to, statements relating to any of our clinical trials, statements relating to the potential benefits of aficamten or any of our other drug candidates, or our ability to obtain regulatory approval for aficamten in any jurisdiction by any particular date, if ever. Cytokinetics' research and development activities; the design, timing, results, significance and utility of preclinical and clinical results; and the properties and potential benefits of Cytokinetics' other drug candidates. Such statements are based on management's current expectations, but actual results may differ materially due to various risks and uncertainties, including, but not limited to, potential difficulties or delays in the development, testing, regulatory approvals for trial commencement, progression or product sale or manufacturing, or production of Cytokinetics' drug candidates that could slow or prevent clinical development or product approval; Cytokinetics' drug candidates may have adverse side effects or inadequate therapeutic efficacy; the FDA or foreign regulatory agencies may delay or limit Cytokinetics' ability to conduct clinical trials; Cytokinetics may be unable to obtain or maintain patent or trade secret protection for its intellectual property; standards of care may change, rendering Cytokinetics' drug candidates obsolete; and competitive products or alternative therapies may be developed by others for the treatment of indications Cytokinetics' drug candidates and potential drug candidates may target. For further information regarding these and other risks related to Cytokinetics' business, investors should consult Cytokinetics' filings with the Securities and Exchange Commission.
CYTOKINETICS® and the CYTOKINETICS and C-shaped logo are registered trademarks of Cytokinetics in the U.S. and certain other countries.
Contact:
Cytokinetics
Diane Weiser
Senior Vice President, Corporate Affairs
(415) 290-7757
References:
- Hegde, SM. Effect of Aficamten Compared with Metoprolol on Echocardiographic Measures in Symptomatic Obstructive Hypertrophic Cardiomyopathy: MAPLE-HCM. JACC. 2025. DOI: .
- Rowin, EJ, et al. Low Incidence of Atrial Fibrillation in Patients with Obstructive HCM Treated with Aficamten: An Analysis from the REDWOOD-HCM, SEQUOIA-HCM and FOREST-HCM Trials. Heart Rhythm. 2025.
- Chuang C, Collibee S, Ashcraft L, et al. Discovery of Aficamten (CK-274), a Next-Generation Cardiac Myosin Inhibitor for the Treatment of Hypertrophic Cardiomyopathy. J Med Chem. 2021;64(19):14142��14152. https://doi.org/10.1021/acs.jmedchem.1c01290
- CVrg: Heart Failure 2020-2029, p 44; Maron et al. 2013 DOI: 10.1016/S0140-6736(12)60397-3; Maron et al 2018 10.1056/NEJMra1710575
- Symphony Health 2016-2021 Patient Claims Data DoF;
- Maron MS, Hellawell JL, Lucove JC, Farzaneh-Far R, Olivotto I. Occurrence of Clinically Diagnosed Hypertrophic Cardiomyopathy in the United States. Am J Cardiol. 2016; 15;117(10):1651-1654.
- Gersh, B.J., Maron, B.J., Bonow, R.O., Dearani, J.A., Fifer, M.A., Link, M.S., et al. 2011 ACCF/AHA guidelines for the diagnosis and treatment of hypertrophic cardiomyopathy. A report of the American College of Cardiology Foundation/American Heart Association Task Force on practice guidelines. Journal of the American College of Cardiology and Circulation, 58, e212-260.
- Hong Y, Su WW, Li X. Risk factors of sudden cardiac death in hypertrophic cardiomyopathy. Current Opinion in Cardiology. 2022 Jan 1;37(1):15-21
A photo accompanying this announcement is available at








